X-Ray Fluorescence spectrometry involves the measurement of emission lines observed when a material is bombarded with X-Rays. Because elements differ in the energy of the lines, and intensity is proportional to concentration, XRF is useful in both qualitative and quantitative inorganic analysis.
In a highly simplified explanation of this phenomenon, when an X-Ray photon of sufficient energy collides with an atom, it may be absorbed and result in the ejection of an inner shell electron, typically in the K or L shells (principal quantum numbers, n, of 1 or 2, respectively). The resulting ionized atom relaxes from an excited state via transition of an electron from one of the higher shells (e.g., L, M) to fill the gap left by the ejected electron. This results in the emission of a photon whose energy corresponds to the difference in binding energies of the two shells. The overall process of emission of radiation of a wavelength that is different from the one absorbed is called fluorescence. The origin of emission lines in an XRF spectrum is illustrated in the figure below.
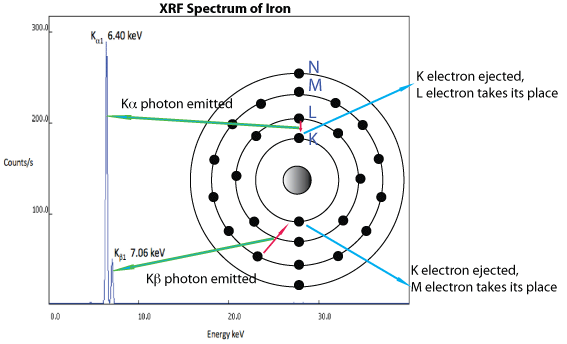
The resulting X-Ray photons can be measured by a detector and the data plotted as intensity (counts/s) as a function of energy (keV). The pattern or fingerprint of lines for a material may be deconvoluted by using one of several software algorithms to estimate % composition for the elements in the material tested. An overview of XRF spectrometry as utilized by by an analyzer is illustrated below.
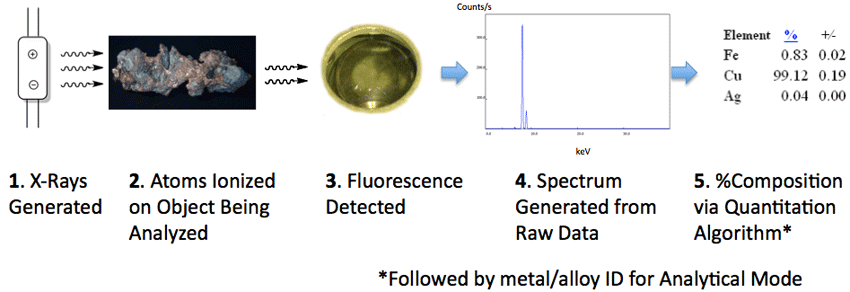
The quantitation algorithms used to determine % composition from the element line intensities include: (1) Fundamental Parameters (2) Compton Normalization and (3) Empirical Calibrations.
For more detailed information on X-Ray Fluorescence spectrometry, see: XRF Data under Resources.